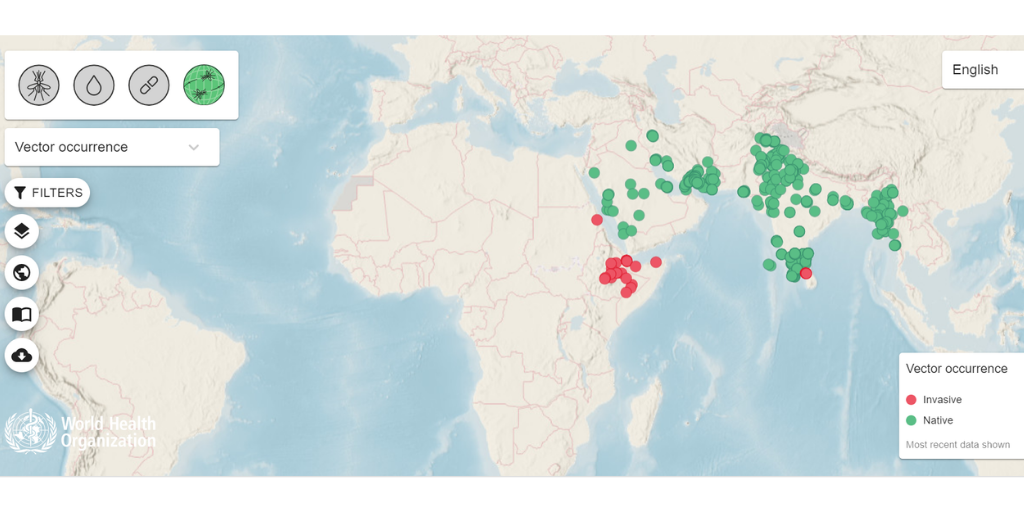
There are signs that an Indian mosquito called Anopheles stephensi, is invading Africa. But why does this matter?
Africa already has the world’s most efficient malaria vectors (namely Anopheles gambiae and An. funestus), and as a result, it has 85% of the world’s malaria deaths. By comparison, Anopheles stephensi is only moderately efficient in carrying the parasite from one person to another; as a vector, this is not bad by global standards, but not nearly as good as the African species. So, is it a big deal if the Indian species invades and becomes established alongside the African species that are already present? Obviously, the addition of an extra vector species is probably not a good thing, but will it really make a vast difference to the situation as a whole?
The answer is: yes, this is a major threat to the region.
Insecticide resistance is currently the most urgent and dangerous threat to recent progress in malaria control across Africa, but the arrival of An. stephensi is a new and almost equally important threat. This is because An. stephensi is uniquely well-adapted to urban conditions.
African cities such as Kigali, Dakar and Nairobi are expanding rapidly, as people are driven to migrate by powerful forces: not only rural poverty and the search for economic opportunity, but also landscape degradation and climate change. Already, some 40% of the African population is urban, and this proportion is expected to rise to 60% by 2050. However, An. gambiae and An. funestus, like most Anopheles species, cannot breed in small containers, or in water with organic pollution, and therefore these mosquitoes tend to be excluded by the process of urbanisation. As a result, African towns typically have fewer mosquitoes and less malaria than surrounding rural areas. The centres of large African cities can be completely free of malaria transmission, and in some countries, these are the only places that are reliably free of transmission. Thus, while many health problems are exacerbated by the ongoing process of urbanisation, malaria is one problem that tends to be “built out” as housing improves and the landscape gradually becomes more and more polluted and paved over with concrete.
Indian cities, by contrast, have An. stephensi, which breeds abundantly in water-storage containers of all kinds, and is the only important malaria vector that is well-adapted to urban areas. India is therefore the only part of the world where the process occurs backwards, with malaria routinely exported from cities to the surrounding villages.
What are the consequences?
If An. stephensi spreads into cities throughout tropical Africa, the consequences are likely to be serious. First, intense malaria could spread to the inner-city areas that, until now, have had little or no transmission. Second, we will no longer be able to assume that in the long run, the process of urban development is inexorably bringing us closer to the goal of malaria elimination. We may find that when we build new suburbs, we are effectively building malaria into the landscape, not building it out. In short, if An. stephensi does spread through urban Africa, it will make a bad situation much worse.
Can we learn from previous invasive vectors?
In the 1930s, An. gambiae invaded Brazil. The world was slow to react, but eventually recognised that in doing nothing, it would spread throughout the continent, and the consequences would be truly catastrophic. The scientific community mobilised and started a coordinated campaign with international support. This was before the advent of DDT and other synthetic insecticides, and required vigorous, disciplined, and sustained efforts. But in the end, An. gambiae was completely eradicated in Brazil. This is one of the big success stories of pre-WW2 public health. If it had failed, or delayed too long, then for the last 80 years, we might have seen African levels of malaria transmission intensity in much of South and Central America. The costs are hard to estimate, but would certainly include tens of millions of additional deaths and many billions of additional dollars for sustained malaria control interventions.
Of course, eradicating An. stephensi from Africa would be difficult and expensive. But it would almost certainly be easier than it was to eradicate An. gambiae from Brazil. It would also be much cheaper, in the long run, than leaving the new species to spread to towns and cities throughout Africa, and then spend many years trying to suppress it locally, using interventions that are expensive and only partially effective. This is an “emerging infectious disease” disaster that we can still prevent – but only if we act decisively now.
If you enjoyed this article and would like to build a career in global health, we offer a range of MSc programmes covering health and data, infectious and tropical diseases, population health, and public health and policy.
Available on campus or online, including flexible study that works around your work and home life, be part of a global community at the UK's no.1 public health university.