Much interest has emerged recently about the potential role that chloroquine, a well-known anti-malarial agent, may play in the treatment of the novel coronavirus (SARS-CoV-2), the causative agent of the current COVID-19 pandemic. Both Italy and France have recently permitted the prescribing of a related drug, hydroxychloroquine, for treating COVID-19 infection. But does the existing evidence support the re-purposing of this old malaria drug for a new viral scourge?
Chloroquine as an anti-malarial agent
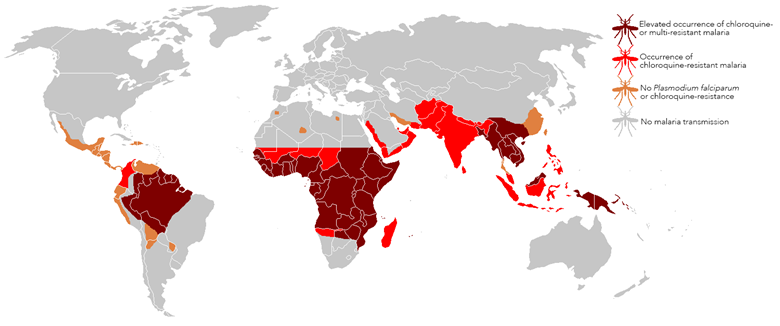
Since its discovery and development in the 1940s, chloroquine has been disseminated across the world to treat all species of Plasmodium – the parasite causing malaria. It was highly valued for its potency against malaria parasites, but also for being safe, cheap to produce, and stable in tropical conditions. It was so safe and stable that it was added to table salt, in regions where malaria was endemic, to enhance malaria elimination efforts at that time. This and other indiscriminate uses had their consequences.
In the late 1950s and early 1960s, resistance to chloroquine began to emerge in South East Asia and South America to Plasmodium falciparum, the malaria species causing most infections and mortality. By the end of the 20th century, chloroquine resistance in this species had spread across the world and drug treatment failures were costing lives. The World Health Organisation (WHO) stopped recommending chloroquine for Plasmodium falciparum malaria (replacing it with Artemisinin-based Combination Therapies), but chloroquine continues to be recommended as treatment for other malaria species, in areas where there is little or no evidence of resistance.
Chloroquine as an anti-inflammatory
Chloroquine, and a related drug hydroxychloroquine (not used as an anti-malarial), have also been shown to exhibit anti-inflammatory properties. Both drugs are now being used to treat autoimmune diseases such as lupus erythematosus and rheumatoid arthritis.
Chloroquine as an antiviral agent
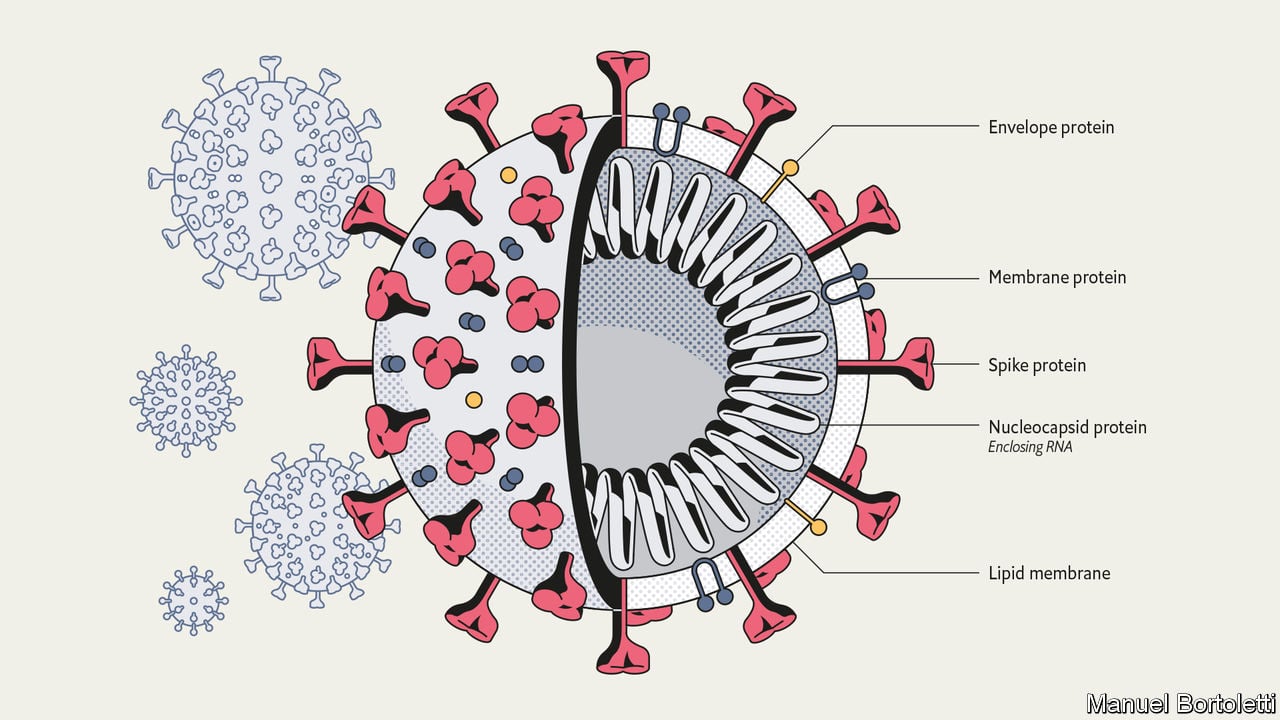
SARS-CoV-2, the virus causing the pandemic COVID-19 disease, belongs to a family of viruses, collectively referred to as coronaviruses. Another coronavirus, SARS-CoV-1, was responsible for the SARS epidemic earlier this century (2002-2003). In 2003 researchers proposed that chloroquine might be useful to prevent the invasion and growth of SARS-CoV-1, as it had been shown previously with other viruses. Then, in 2005 the first evidence emerged showing that chloroquine could indeed prevent the invasion and spread of SARS-CoV-1 coronavirus in primate cells in the laboratory. However, the following year a further study in a mouse model showed chloroquine was not effective at clearing a SARS-CoV-1 infection.
Recently, there has been a revival in research into chloroquine and coronaviruses. Studies are emerging from China showing that chloroquine can inhibit SARS-CoV-2 infection in the laboratory, and suggesting how it might be used to treat COVID-19 infection. A recent review lists a number of human trials in China designed to test the efficacy of chloroquine against the COVID-19 disease, but these trials are only likely to begin showing outcomes a few months from now. Recently, a French trial on a small number of patients showed that hydroxychloroquine may be useful in treating COVID-19. However, this trial has been criticised for its small patient numbers and poor trial design, calling into question their conclusions.
Optimal dosing regimen: risk of toxicity at high doses?
Evidence is therefore mounting that chloroquine and hydroxychloroquine are able to inhibit coronavirus infection in the laboratory. However, a crucial question remains: are these drugs going to be effective in humans? A major concern is that some of the doses needed to clear viral infections in the laboratory will be too high for human use and may result in drug toxicity.
Current Chinese trials testing chloroquine for COVID-19 treatment explore a range of concentrations and formulations. Some involve between 500 mg to 1 gram of chloroquine per day.
The British National Formulary – an expert guidance list of medicines for the UK – recommends a daily dose of 155 mg chloroquine for treating autoimmune disease. For treating malaria, there is an initial dose of 620 mg, followed by three doses of 310 mg over the following three days. These doses are lower than some of those being trialled in China and it is critical to ascertain whether the higher doses result in toxic effects.
For hydroxychloroquine, the British National Formulary recommendation is 200-400 mg daily for autoimmune conditions.
However, in a coronavirus study underway in France, on only 26 patients receiving the treatment, 600 mg of hydroxychloroquine was used per day; a dose that is 1.5 – 3 times higher than is recommended in the UK for treating autoimmune conditions.
The studies being carried out must answer the important questions of: (1) what is the optimal dose and timing of chloroquine or hydroxychloroquine administration to prevent viral infection and/or disease progression; (2) is this dose achievable without harming the patients; (3) what other drugs should it be combined with to achieve its optimal effect and are these combinations safe?
Dangers of self-medication
In malaria-endemic regions, many people still have access to chloroquine and associate it with being safe to use. This may lure them into a false sense of security regarding the use of this drug to treat COVID-19. Since chloroquine has not been specifically licensed for the purpose of treating COVID-19, its dosing regimen will be for malaria infection. This may be insufficient to effectively suppress or kill the virus. Conversely, self-medication using a higher dose may inadvertently result in chloroquine toxicity (high doses of which can be fatal).
Many answers remain about the optimal use and safety of chloroquine and hydroxychloroquine to treat COVID-19 infections. Without these answers, treating with the current high doses recommended potentially risk exacerbating an already harmful and deadly disease. Should we take that risk in these already vulnerable patients? Or should we wait until the results of definitive clinical trials, now being carried out, are known?
This is a quickly evolving situation, all information is accurate as of Monday 30 March.
Thanks to Dr Don Van Schalkwyk, Dr Colin Sutherland, Prof Jimmy Whitworth & Prof Sian Clarke for their contributions. If you have any questions, please get in touch with [email protected]
If you enjoyed this article and would like to build a career in global health, we offer a range of MSc programmes covering health and data, infectious and tropical diseases, population health, and public health and policy.
Available on campus or online, including flexible study that works around your work and home life, be part of a global community at the UK's no.1 public health university.