Open data sharing: the next strike in the fight against antimicrobial resistance
London School of Hygiene & Tropical Medicine https://lshtm.ac.uk/themes/custom/lshtm/images/lshtm-logo-black.png Tuesday 12 September 2023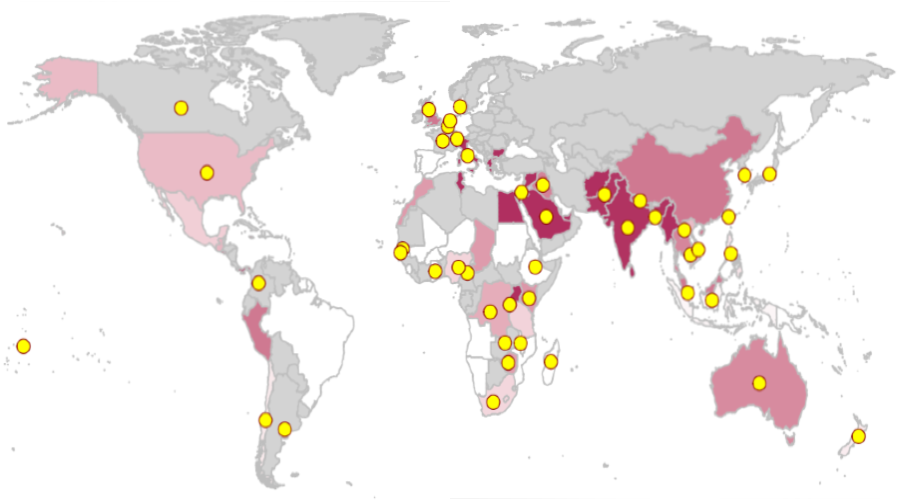
In many parts of the world where people have consistent access to safe water and improved sanitation, typhoid fever is a thing of the past. However, Salmonella Typhi (S. Typhi) still poses a major public health problem in many lower- to middle- income countries, with over 9 million cases and 110,000 deaths estimated in 2019.
Typhoid fever is typically transmitted through the consumption of faecally-contaminated water. As over 2 billion people rely on faecally-contaminated water sources worldwide, this means that almost a third of the global population is at risk. Antimicrobials have been used to treat typhoid for decades, beginning with chloramphenicol in the late 1940s, but with every introduction of a new antimicrobial agent, antimicrobial resistance (AMR) has followed.
The emergence of an extensively drug-resistant S. Typhi variant in Pakistan, resistant to every oral antimicrobial used to treat typhoid fever except azithromycin, made international news headlines in 2016. Since then, there have been reports of resistant variants found in Bangladesh, Pakistan, India, and Nepal, adding to growing concerns about the looming threat of “untreatable” typhoid fever.
Set against this backdrop, the Global Typhoid Genomics Consortium (GTGC) was established to bring together the global typhoid research community to combine as much S. Typhi genomic data as possible to inform public health action. Our first objective was to provide an updated overview of global genotype and AMR distribution and trends over time. Twenty-two years on from the publication of the first S. Typhi genome, our latest analysis, which has recently been published in eLife, includes 13,000 S. Typhi genomes from across 110 countries and is the largest published collection to date.
While previous genomic studies of typhoid have focused on a specific pathogen lineage or variant, combining data from different countries has highlighted local variants that have evolved independently to be equally drug-resistant. Whether these variants can spread to neighboring countries, or whether they have already done so, is unknown in many regions and should be monitored with continued and expanded genomic surveillance. We also found that a current lack of widespread surveillance may mean that certain antimicrobials, such as ciprofloxacin, are being used in settings where resistance is already likely present but undetected.
The open-collaboration model of our study is quite unique in the world of bacterial genomics research. Consortium membership is open to anyone interested and engaged in typhoid surveillance and sequencing, and we welcome all researchers in the field globally. For maximum impact and reproducibility, data included in our analysis have been deposited in public archives and our analysis has been conducted using open-access tools and publicly-available code.
It’s hoped that by continuing to monitor the emergence and global spread of AMR S. Typhi, our work can inform decisions concerning the introduction of typhoid conjugate vaccines and other preventative interventions, such as improved water and sanitation. This serves as an exciting starting point for expanded genomic data sharing and collective analysis, building on the momentum of the research community’s response to the COVID-19 pandemic.
LSHTM's short courses provide opportunities to study specialised topics across a broad range of public and global health fields. From AMR to vaccines, travel medicine to clinical trials, and modelling to malaria, refresh your skills and join one of our short courses today.