Spotlight on...Plasmodium falciparum: Genesis or demise
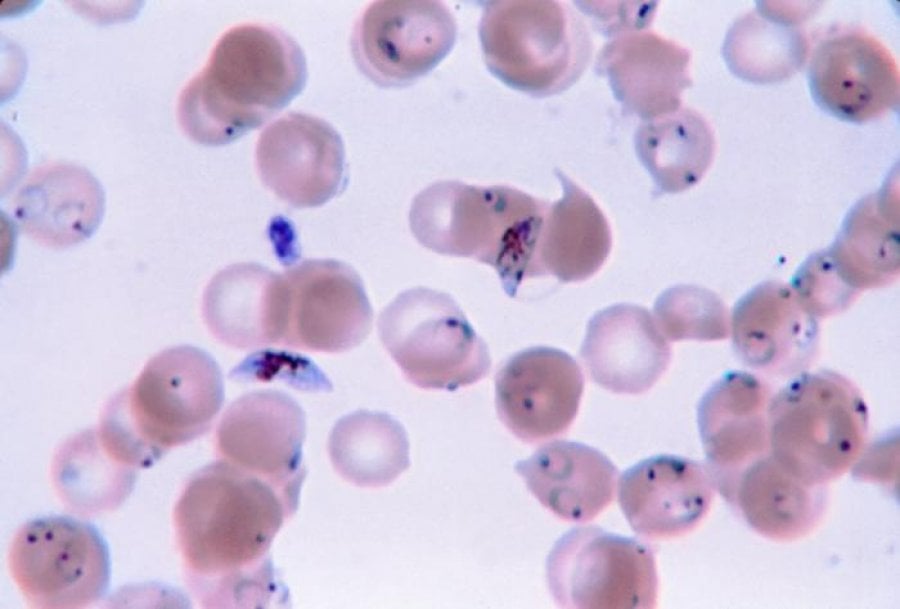
Malaria parasites rely on the sexually differentiated gametocytes to transmit the malaria parasite from the mammalian host to the biting mosquito. Interventions that specifically prevent human-to-mosquito transmission by targeting gametocytes are key to elimination.
We are delighted to present a fantastic line-up of speakers who will focus on different aspects of Plasmodium falciparum gametocytes, which differ from other species of medical importance in their extended circular time, elongated structure, and distinct patterns of drug susceptibility.
- Dr Gillian Stresman (Chair)
-
Gillian Stresman is an Assistant Professor at the London School of Hygiene & Tropical Medicine. She has been working on malaria epidemiology research for more than 10 years and has spent time in multiple malaria-endemic countries including Zambia, Kenya, Philippines and Haiti. Her focus is on understanding the epidemiological and spatial determinants of malaria transmission with the ultimate goal of accelerating transmission reduction and achieving elimination. This includes studies ranging from understanding bias in routine surveillance due to asymptomatic infections, identifying residual parasite populations to target interventions, understanding the role of spatial transmission dynamics at various spatial-scales, and identify tools and metrics for malaria surveillance that are operationally feasible for implementation in endemic settings. Additional research focuses on malaria elimination in terms of both how to achieve zero as well as how to measure absence of infections.
- Catherine Lavazec, PhD, Biology of Plasmodium transmission team, Institute Cochin (CNRS), University of Paris
-
Title:
Plasmodium falciparum sexual development in the human bone marrow
Bio:
Dr Catherine Lavazec is a Research Director and Principal Investigator at Institut Cochin (CNRS) in Paris. During her PhD and post-doctoral work at Institut Pasteur in Paris and Cornell University in New York, she contributed to the characterization of several Plasmodium and Anopheles proteins families involved in parasite transmission. In 2013, she joined the Institut Cochin where her research is focused on unique biology underpinning Plasmodium falciparum transmission and the cellular mechanisms driving this process. The Lavazec lab aims to decipher the interactions between P. falciparum gametocytes and their microenvironment, particularly in the bone marrow and the spleen, as well as the changes in red blood cells mechanical properties during gametocyte maturation. The lab has identified parasite proteins and signaling pathways that participate to these processes and has established that these mechanisms may be new targets to block malaria parasite transmission.
-
Title:
Plasmodium falciparum sexual development in the human bone marrow
Bio:
Dr Catherine Lavazec is a Research Director and Principal Investigator at Institut Cochin (CNRS) in Paris. During her PhD and post-doctoral work at Institut Pasteur in Paris and Cornell University in New York, she contributed to the characterization of several Plasmodium and Anopheles proteins families involved in parasite transmission. In 2013, she joined the Institut Cochin where her research is focused on unique biology underpinning Plasmodium falciparum transmission and the cellular mechanisms driving this process. The Lavazec lab aims to decipher the interactions between P. falciparum gametocytes and their microenvironment, particularly in the bone marrow and the spleen, as well as the changes in red blood cells mechanical properties during gametocyte maturation. The lab has identified parasite proteins and signaling pathways that participate to these processes and has established that these mechanisms may be new targets to block malaria parasite transmission.
- Prof Alassane Dicko, Professor of Epidemiology and Head of Clinical Research Unit of Oulessebougou-Bougouni, Malaria Research and Training Center, University of Science Techniques and Technologies of Bamako
-
Title :
Field studies in Mali of Low single dose of Primaquine to block falciparum malaria transmission.Bio :
Professor of Epidemiology and Public Health at Malaria Research and Training Center (MRTC) of the University of Science Techniques and Technologies of the Bamako and a Senior Investigator at the Bruyère Research Institute of the University of Ottawa. He holds an MD from the University of Bamako, a Master of Science in Epidemiology from the University of Maryland and a PhD in Epidemiology from the University of Bordeaux, France.
His research has focused on improving ways of preventing and treating malaria in areas where malaria transmission is highly seasonal, such as Mali. He has been a pioneer in the development of Seasonal Malaria Chemoprevention, a control measure now being deployed widely in many countries of the Sahel and sub-Sahel where malaria transmission is highly season. With colleagues at the London School of Hygiene and Tropical Medicine and the University of California San Francisco, the US National Institutes of Health and USAID he is monitoring the implementation of SMC in Mali and other Sahelian countries and testing strategies to improve it through addition transmission blocking drugs such as low single dose of primaquine and or seasonal vaccination with RTSS and PfSPZ.
He also tested the safety and efficacy of low single dose of primaquine in blocking malaria transmission in G6PD deficient and non-deficient subjects and have shown that a single 0·25 mg/kg dose of primaquine is effective in blocking the transmission of P. falciparum in mosquitoes using membrane feeding assay and that Methylen Blue is also effective in blocking malaria transmission of P. falciparum in mosquitoes using membrane feeding assay.
He has initiated and developed two research field sites in Ouelessebougou and Bougouni in Mali with trained staff and state of art equipment to conduct Phase 1, 2 and 3 vaccines and drug trials. In 2017, he was awarded the prestigious Royal Society’s Africa prize for his work on the prevention of malaria in African children. Dicko has coauthored more than 100 peer review publications including in high impact journals such as Lancet and NEJM.
- Dr Michael Delves, Assistant Professor, Department of Infection Biology, LSHTM
-
Title:
"Characterising promising new transmission-blocking antimalarials through phenotypic imaging and machine learning analysis"Bio:
Michael studied Biochemistry at Imperial College before completing a PhD in Cellular and Molecular Neurobiology in 2008. He then moved into the field of malaria transmission research in the Lab of Prof Bob Sinden, playing an instrumental part in establishing the Medicines for Malaria Venture Centre of Excellence at Imperial and developing gold standard high throughput assays for the discovery of new transmission-blocking antimalarial drugs.Michael moved to LSHTM in August 2018 to begin his independent career focusing on Plasmodium falciparum transmission cell biology.
Please note that the time listed is Greenwich Mean Time (GMT)
Admission
Contact




