Wolbachia - has the world a new weapon to fight dengue fever?
London School of Hygiene & Tropical Medicine https://lshtm.ac.uk/themes/custom/lshtm/images/lshtm-logo-black.png Friday 3 August 2018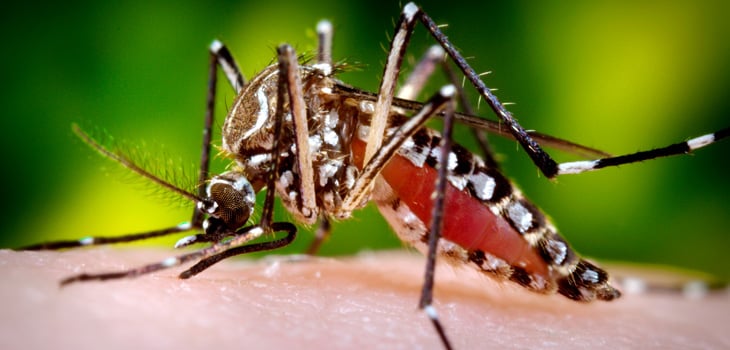
Currently there are no therapeutic drugs against the disease and existing licensed vaccines are only partially effective and have complex eligibility requirements. As a result, efforts to stop dengue have been targeted at Aedes aegypti - its day-biting, urban dwelling mosquito vector.
Proving any vector control tool works against dengue is a considerable challenge. While many vector control tools can show that they can kill mosquitoes, there are many reasons why this may not translate to reductions in dengue cases due to the complex immunology and high transmission intensity of the disease
While many can show that they can kill mosquitoes, there are several reasons why this may not translate to reductions in dengue cases due to the complex immunology and heterogeneity of the disease.
Very few vector control trials with epidemiological endpoints have ever been conducted, and those that have, show variable results. This is why the first long-term results of the Wolbachia release programme in northern Australia, unveiled this week, have been so eagerly anticipated.
It details the use of a highly novel approach where instead of killing mosquitoes, the population is entirely replaced with mosquitoes infected with an intracellular bacterium called Wolbachia that blocks passage of the dengue virus. It is also the first time that this technology has been shown to be durable over multiple years and can be deployed at scales that can have a meaningful impact on dengue incidence.
Since the introduction of Wolbachia mosquitoes in Townsville, northern Australia, the regular bi-annual cycle of outbreaks appears to have stopped. Of course, there are many reasons why this might have happened, but absence of outbreaks despite repeated introduction of dengue through infected returning travellers, gives us more confidence that Wolbachia is causing this effect.
This interrupted timeseries type trial design with no clear ‘control’ group is short of the kind of evidence we would like to have, and indeed would require for licensure of a drug or vaccine. But there is an active ongoing discussion about the standard of evidence required for interventions that are deployed at a community (instead of individual) level.
The results released this week are just one further block in building the evidence base for the effectiveness of Wolbachia. Questions still remain about how long this intervention will remain effective, or whether the reduction in transmission potential is enough to have a meaningful impact on cases in the highest transmission settings in Asia and Latin America.
Ongoing monitoring of Wolbachia coverage and effectiveness occurs in all release sites, and the stability of Wolbachia infected mosquitoes for seven years in Townsville is an encouraging result. A more traditional cluster randomised trial for Wolbachia is currently underway in Yogyakarta, Indonesia with results expected in the next 2-3 years.
But for many this wait is too long. The emergence of Zika virus (which Wolbachia is also effective against) has accelerated the scale-up of Wolbachia releases with city-wide programmes in Medellin and Rio de Janeiro that aim to protect millions of people.
The next phase will test the scalability of this intervention. This method relies on strong community engagement in addition to considerable financial investment to conduct the releases, neither of which are trivial to achieve at these scales. Model-based predictions of the costs and benefits of scale up of Wolbachia can help make the case for optimal investment in the next stage of the programme.
Despite the remaining uncertainties, when billions of people are at risk and with limited other options, there will be considerable pressure on seeing this promising new technology scaled-up without delay.
Our postgraduate taught courses provide health practitioners, clinicians, policy-makers, scientists and recent graduates with a world-class qualification in public and global health.
If you are coming to LSHTM to study a distance learning programme (PG Cert, PG Dip, MSc or individual modules) starting in 2024, you may be eligible for a 5% discount on your tuition fees.
These fee reduction schemes are available for a limited time only.
